Learn about common corrosion issues in canted coil springs—including pitting, stress corrosion cracking, and galvanic corrosion—and discover proven prevention strategies through material selection and plating for reliable performance in harsh environments.
Canted coil springs are precision components that play a critical role in sealing, EMI shielding, electrical contacts, and mechanical latching across industries such as semiconductor equipment, aerospace, medical devices, and oil and gas. Their unique canted geometry delivers near-constant force over a wide deflection range, making them indispensable in demanding applications.

However, these springs often operate in harsh environments—exposed to moisture, salt spray, chemicals, or temperature extremes. Corrosion is the single most common cause of premature spring failure, leading to force decay, increased contact resistance, loss of shielding effectiveness, and ultimately system malfunction.
This article examines the specific corrosion threats facing canted coil springs, explains how and why different materials behave under corrosive attack, and provides practical strategies to prevent corrosion through informed material selection and surface treatment.
Canted coil springs are manufactured from metal strips or wires that are precision-formed, heat-treated, and often plated. Unlike a simple structural component, a spring is designed to store and release mechanical energy through elastic deformation. This makes it particularly sensitive to surface damage.
Corrosion initiates at the surface. Even minor pitting or localized attack can create stress concentration points that act as crack initiation sites under cyclic loading. Once a crack starts, fatigue propagation is accelerated, leading to premature fracture. Moreover, corrosion products on contact surfaces increase electrical resistance, compromising EMI shielding effectiveness and signal integrity.
Canted coil springs typically experience sustained mechanical stress during service. The combination of tensile stress and a corrosive environment creates the perfect conditions for stress corrosion cracking (SCC), one of the most dangerous and insidious forms of corrosion attack.
Understanding the specific corrosion mechanisms that threaten canted coil springs is the first step toward effective prevention.
Pitting is a localized form of corrosion that produces small cavities or “pits” on the metal surface. It is particularly dangerous because pits are difficult to detect visually and can penetrate deep into the material while the surrounding surface appears unaffected.
Causes: Pitting typically occurs in materials that rely on a passive oxide layer for protection, such as stainless steels. Chloride ions (from salt water, de-icing chemicals, or industrial processes) break down the passive layer locally, creating an electrochemical cell that rapidly corrodes the exposed metal beneath.
Symptoms in springs: Reduced cross-sectional area at the pit site leads to localized stress concentration, reduced spring force, and eventual fracture under cyclic loading.
Crevice corrosion occurs in shielded areas where stagnant solutions accumulate. For canted coil springs installed in grooves, the interface between the spring and the groove wall creates a natural crevice.
Causes: Oxygen depletion within the crevice creates a differential aeration cell, accelerating corrosion inside the crevice while the external surface remains protected.
Symptoms in springs: Crevice attack typically occurs where the spring contacts the housing, leading to loss of contact integrity, increased electrical resistance, and eventual loss of spring function.
Galvanic corrosion occurs when two dissimilar metals are in electrical contact in the presence of an electrolyte (such as moisture). The more active metal corrodes at an accelerated rate, while the more noble metal is protected.
Causes: In many canted coil spring applications, the spring is made of stainless steel or beryllium copper while the housing or mating surface is aluminum. Aluminum is significantly more active than either stainless steel or copper alloys, creating a strong galvanic couple. When moisture is present, aluminum corrosion accelerates dramatically.
Symptoms in springs: The spring itself may show little corrosion, but the aluminum housing around it will suffer rapid pitting and material loss, loosening the spring and destroying the seal.
Stress corrosion cracking is the combined action of sustained tensile stress and a corrosive environment. It produces brittle cracks that propagate through the material, often with little or no visible warning.
Causes: Canted coil springs are under constant mechanical stress by design. When combined with a specific corrosive medium (chlorides for stainless steels, ammonia for copper alloys, etc.), SCC can initiate at stress concentrations and propagate rapidly.
Symptoms in springs: Sudden, unexpected fracture without significant general corrosion visible on the surface.
Corrosion fatigue is similar to SCC but occurs under cyclic rather than static loading. The spring experiences alternating stresses (such as vibration or repeated compression/relaxation cycles) while exposed to a corrosive environment.
Causes: Corrosive attack accelerates crack initiation, and cyclic loading drives crack propagation. The combination reduces fatigue life by orders of magnitude compared to operation in a non-corrosive environment.
Symptoms in springs: Premature fracture after fewer cycles than expected. A train spring failure analysis revealed that water vapor and corrosive ions penetrating an organic coating produced corrosive pits that became stress concentration points, leading to corrosion fatigue fracture under cyclic loading.
While less common for the high-alloy materials typically used in canted coil springs, uniform corrosion can occur in less noble materials exposed to aggressive environments. The entire exposed surface corrodes at a relatively uniform rate, gradually reducing cross-section and spring force.
Causes: Prolonged exposure to acids, alkalis, or other corrosive media that attack the base material uniformly.
Symptoms in springs: Gradual force decay over time, eventual loss of function.
Prevention requires a systematic approach: understanding the environment, selecting the right base material, applying appropriate surface treatments, and implementing sound design practices.
Material selection is the single most effective corrosion prevention measure. The table below summarizes the corrosion resistance characteristics of common canted coil spring materials:
Detailed material guidance:
301 Stainless Steel: Offers high strength after cold working and is cost-effective for high-volume production, but has lower corrosion resistance than 304 or 316 and is not suitable for marine or chloride-rich environments.
304 Stainless Steel: Provides excellent corrosion and oxidation resistance, suitable for clean and humid environments, making it ideal for medical devices, food-grade equipment, and EMI shielding applications.
316 Stainless Steel: Contains molybdenum, which provides superior corrosion resistance—especially against chlorides. It is the best stainless steel choice for marine equipment, chemical processing, and outdoor EMI shielding where salt exposure is a concern.
Beryllium Copper: Offers higher resistance to corrosion and oxidation than steel, excellent durability in atmospheric, marine, and mild acid/alkali environments. It is ideal for offshore and chemical applications.
Elgiloy®: One of the best all-around alloys for corrosion resistance. Resistant to sour gas environments per ISO 15156-3:2015 (NACE) and much less likely to display hydrogen embrittlement. A top choice within chemical processing and oil and gas industries.
Hastelloy® C-276: Offers outstanding resistance to pitting, crevice corrosion, and stress corrosion cracking. Ideal for highly corrosive environments, extreme marine applications, and chlorine atmospheres.
Inconel®: Known for high temperature strength and corrosion resistance, preferred for extreme environments including cryogenic and high-temperature applications in the chemical processing industry.
When the base material alone does not provide sufficient corrosion resistance, or when galvanic compatibility with mating materials is a concern, plating is the solution.
Common platings for canted coil springs include:
| Plating | Benefits | Best For | Limitations |
|---|---|---|---|
| Tin | Good conductivity, solderable, low cost | General EMI shielding, cost-sensitive applications | Soft, limited wear resistance |
| Nickel | Hardness, wear resistance, good corrosion resistance | Base layer for other platings, industrial environments | Nickel layer has extremely high hardness and wear resistance and good corrosion resistance |
| Silver | Highest conductivity, good oxidation resistance | High-frequency EMI shielding, high-reliability contacts | Expensive, prone to tarnish (but remains conductive) |
| Gold | Excellent corrosion resistance, biocompatible | Medical devices, aerospace, critical contacts | Very expensive |
Gold, silver, nickel, and tin platings are all available for optimal conductivity, galvanic compatibility, and corrosion resistance.
Critical note on galvanic compatibility: When dissimilar metals must contact each other, galvanic compatibility is managed by finishes and plating that facilitate the contact and protect base materials from corrosion. For example, if an aluminum housing must contact a stainless steel spring, plating the spring with a metal closer to aluminum on the galvanic series (such as tin or nickel) can significantly reduce the corrosion rate.
Design details dramatically influence corrosion susceptibility:
Avoid moisture entrapment: Grooves should be designed to allow drainage and prevent pooling of liquids. Sharp corners should be avoided as they create crevices that trap moisture.
Control compression: Over-compressing the spring beyond recommended levels increases residual stress, making the spring more susceptible to SCC. Optimal compression is typically 20-30% of free height.
Seal the interface: For applications with moisture exposure, consider adding an environmental seal (such as an O-ring or silicone bead) outside the EMI shielding spring to keep the contact interface dry. Proper installation helps prevent the ingress of moisture and corrosive agents and ensures long-term effectiveness of the shielding.
Surface finish: Mating surfaces should be smooth (Ra ≤ 0.8 μm) and free of burrs that could damage platings and expose base material to attack.
Even with optimal material selection and design, periodic inspection is essential for long-term reliability:
Visual inspection: Check for discoloration, pitting, or corrosion products on spring surfaces and housing contact areas.
Force testing: Measure spring force periodically to detect force decay indicative of corrosion-related section loss.
Electrical testing: Monitor contact resistance. Rising resistance suggests surface oxidation or corrosion.
Regular inspection and maintenance ensure long-term shielding effectiveness and prevent unexpected failure.
When the operating environment can be modified, simple measures dramatically reduce corrosion risk:
Control humidity: Keep equipment in conditioned spaces where relative humidity is below 60% to minimize electrolyte formation.
Avoid dissimilar metal contact: Use isolating washers or coatings to separate the spring from the housing when galvanic couples cannot be avoided.
Apply protective coatings to mating surfaces: Anodizing aluminum housings or applying conversion coatings to other metals reduces their activity and slows galvanic attack.
Use the following flowchart to guide your material selection for canted coil springs based on corrosion risk:
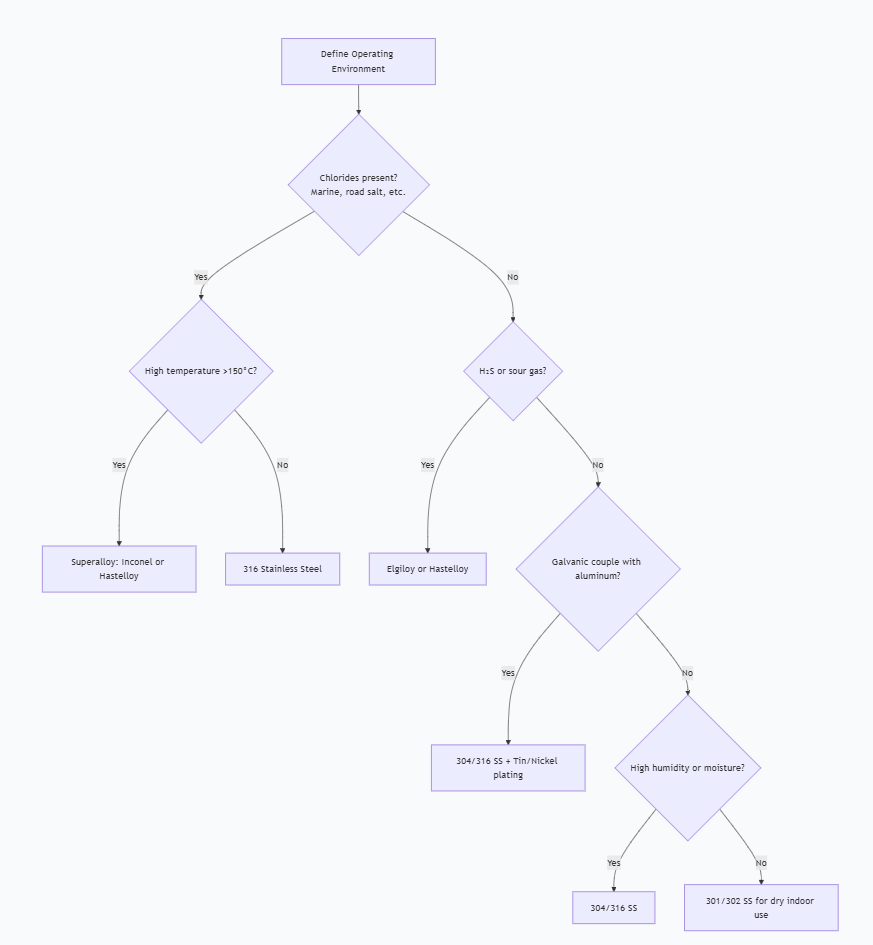
Scenario: A semiconductor wafer fabrication chamber required EMI shielding between an aluminum housing and a stainless steel door. After six months of operation, engineers noticed increasing EMI leakage and intermittent grounding failures.
Diagnosis: Inspection revealed severe pitting corrosion on the aluminum housing around the EMI shielding spring. The galvanic couple between the stainless steel spring (noble) and the aluminum housing (active) in the presence of moisture from the cleanroom environment had accelerated aluminum corrosion.
Solution: The spring was changed from bare 304 stainless steel to 304 stainless steel with electroless nickel plating. Nickel plating is closer to aluminum on the galvanic series, reducing the driving voltage for galvanic corrosion.
Result: After the modification, aluminum pitting was eliminated, and EMI shielding effectiveness remained stable through three years of operation.
While exotic alloys and precious metal platings have higher initial costs, they often provide lower total cost of ownership in corrosive environments.
| Material | Relative Cost | Life Expectancy (Harsh Marine) | TCO Rating |
|---|---|---|---|
| 301 SS | Low | <6 months | Poor |
| 304 SS | Low-Moderate | 1-2 years | Fair |
| 316 SS | Moderate | 3-5 years | Good |
| BeCu + Ni plate | Moderate-High | 5-8 years | Very Good |
| Elgiloy | High | 8-10+ years | Excellent |
| Hastelloy | Very High | 10+ years | Excellent |
Exotic alloys provide more resistance and longevity in specific environments. While more expensive to purchase, this can often be negated by longer life and lower maintenance/replacement costs.
Corrosion in canted coil springs is not inevitable. By understanding the specific corrosion mechanisms that threaten your application, and by systematically selecting base materials and platings that match your environment, you can achieve long-term reliable performance.
The key principles are:
For engineers designing mission-critical systems in semiconductor, medical, aerospace, or industrial applications, the investment in proper corrosion prevention for canted coil springs pays dividends in reliability, reduced downtime, and extended product life.
Need assistance selecting the optimal material and plating for your canted coil spring application? Contact our engineering team for corrosion risk assessment and material recommendations.